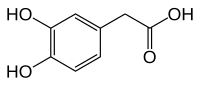
3,4-Dihydroxyphenylacetic acid
 | |
| Names | |
|---|---|
| Preferred IUPAC name (3,4-Dihydroxyphenyl)acetic acid | |
| Other names 2-(3,4-Dihydroxyphenyl)acetic acid | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| DrugBank |
|
| ECHA InfoCard | 100.002.750 |
| KEGG |
|
| MeSH | 3,4-Dihydroxyphenylacetic+Acid |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C8H8O4 |
| Molar mass | 168.148 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
3,4-Dihydroxyphenylacetic acid (DOPAC) is a metabolite of the neurotransmitter dopamine. Dopamine can be metabolized into one of three substances. One such substance is DOPAC. Another is 3-methoxytyramine (3-MT). Both of these substances are degraded to form homovanillic acid (HVA). Both degradations involve the enzymes monoamine oxidase (MAO) and catechol-O-methyl transferase (COMT), albeit in reverse order: MAO catalyzes dopamine to DOPAC, and COMT catalyzes DOPAC to HVA; whereas COMT catalyzes dopamine to 3-MT and MAO catalyzes 3-MT to HVA. The third metabolic end-product of dopamine is norepinephrine (noradrenaline).

It can also be found in the bark of Eucalyptus globulus.[1]
This product has been synthesized (52% yield) from 4-hydroxyphenylacetic acid via aerobic biotransformation using whole cell cultures of Arthrobacter protophormiae.[2][3]
References
- ^ Santos, Sónia A. O.; Freire, Carmen S. R.; Domingues, M. Rosário M.; Silvestre, Armando J. D.; Neto, Carlos Pascoal (2011). "Characterization of Phenolic Components in Polar Extracts of Eucalyptus globulus Labill. Bark by High-Performance Liquid Chromatography–Mass Spectrometry". Journal of Agricultural and Food Chemistry. 59 (17): 9386–93. doi:10.1021/jf201801q. PMID 21761864.
- ^ Robins, Karen T.; Osorio-Lozada, Antonio; Avi, Manuela; Meyer, Hans-Peter (2009). "Lonza: Biotechnology – A Key Ingredient for Success in the Future". CHIMIA International Journal for Chemistry. 63 (6): 327–330. doi:10.2533/chimia.2009.327.
- ^ Sutton, Peter; Whittall, John (2012). Practical Methods for Biocatalysis and Biotransformations 2. Chichester, West Sussex: John Wiley & Sons, Ltd. pp. 150–153. ISBN 9781119991397.
- v
- t
- e
| Anabolism | |||||||
|---|---|---|---|---|---|---|---|
| Catabolism |
|
| Anabolism | |
|---|---|
| Catabolism |
|
- N-Acetylserotonin (NAS; normelatonin)
- 5-Methoxytryptamine (5-MT)
- 5-Methoxyindoleacetaldehyde (5-MIAL)
- 3-Methoxy-4-hydroxyphenylacetaldehyde (HMPAL)
- 4-Hydroxyphenylacetaldehyde (HPAL)
- Indoleacetaldehyde (IAL)
- Phenacetaldehyde (PAL)












